The human body is a problem.
For most of us, it’s a management problem: nutrition, exercise, blood work, the occasional X-ray or MRI, a treatment regimen.
For doctors, it’s a disease problem: What drives the formation of amyloid plaques in Alzheimer’s? Which factors make certain cancers resistant to therapies? Why can’t we detect Parkinson’s sooner? (Doctors trying to study these questions may also say the human body is a funding problem.)
And for researchers in the age of artificial intelligence, the human body is a math problem.
“There are on the order of billions of proteins just in an individual human cell. The three billion nucleotides in the genome specify the entire program for the human body. There’s just tremendous, tremendous complexity,” says Alex Rives, Ph.D., head of science at Biohub, a nonprofit research organization developing AI-powered tools to improve (and speed) human health research.
The complexity becomes exponentially greater once you factor in all the ways disease can arise, explains Rives’s colleague Andrea Califano, Ph.D., Biohub’s president of immune cell reprogramming. “There are 10 to the 400th power possible mutational patterns that can lead to cancer,” he says. “It’s more than the number of atoms in the universe.”
We spoke with four Biohub scientists working at the frontier of this challenge — using new tools, new data, and AI to make the body’s complexity legible.
The missing science of cell behavior
Over the past few decades, scientists have mapped the human genome, identified thousands of cell types, and learned to image living tissue in remarkable detail. Yet a basic question remains unanswered: how do human cells actually behave — moment to moment, under stress, in relation to one another — and how do those behaviors tip the body from health into illness?
In cancer, researchers can identify many of the mutations linked to tumors, but still struggle to predict which altered cells will turn deadly. Two patients with nearly identical tumors can respond very differently to the same treatment, often because of differences in how cells signal, adapt, and interact with their environment in ways we can’t yet reliably observe or control.
In Alzheimer’s and Parkinson’s, pathological proteins accumulate for years or decades before symptoms appear, but the “tipping point” — the moment at which a patient begins to show signs of dementia or disability — remains poorly understood. By the time memory loss or movement problems emerge, irreversible damage has already occurred, leaving clinicians with few options beyond slowing decline.
Simulating life at the cellular level
Because scientists can’t yet observe living human cells as disease unfolds or rewind to the moment something goes wrong at the molecular level, they’re turning to AI to simulate cellular behavior across time and conditions that are difficult to capture in the lab.
“You can think of the human cell as a system of information,” says Rives, a pioneer in AI and biology who developed the first large transformer language models for proteins. “Biology is a kind of language. What would it mean to train a model on the text of life?”
Using massive datasets and scaling up to 10,000 GPUs of compute over the next few years, Rives’s Biohub team is building virtual cells — AI models designed to behave like real human cells, much as language models can behave like a therapist or a math tutor.
“Cells are made up of molecules, they come together in complex circuits, all of this is programmed by the genome, there’s many different layers,” Rives says. “The complexity is far beyond the ability to simulate from physical or chemical first principles. You need an information theoretic centered approach.”
As a step toward building virtual cells, Biohub is scaling up the generation of data. Its Billion Cells Project is an early effort to map the many states a human cell can occupy. Rives believes that training AI on such a map could make it possible to model the underlying mechanisms that govern health and disease. Biohub’s technology development efforts across frontier imaging, instrumentation and measurement are expected to expand both the breadth and resolution of that map over time, giving models a more detailed picture of human biology. Ultimately, Rives’s team aims to leverage AI to speed the search for ways to prevent and cure disease.
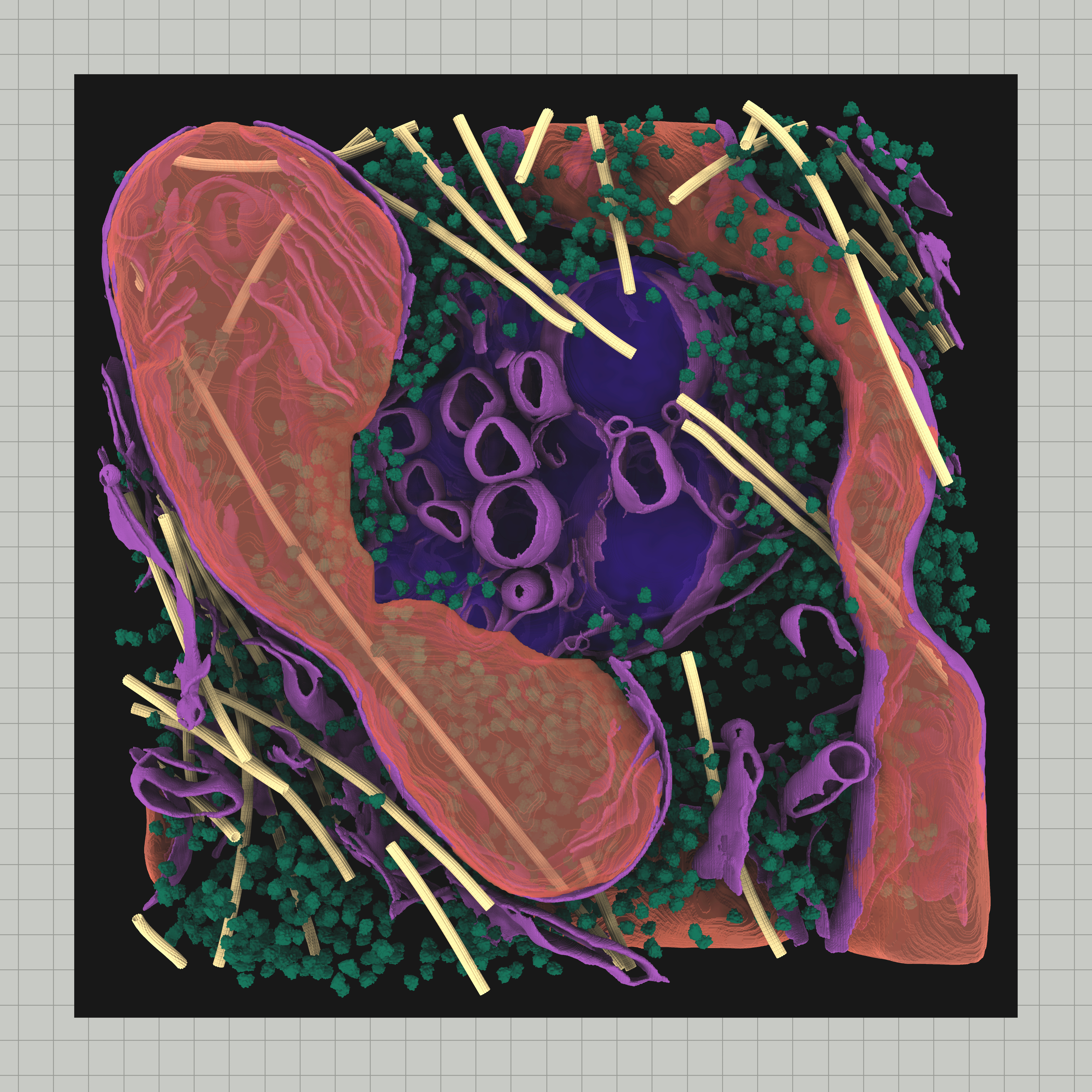
From snapshots to imaging systems
Imagine trying to understand the inner workings of a city from a single aerial photograph. You’d see buildings and one moment of the weather, but not traffic patterns, the power grid, or how the environment changes over time. Modern imaging tools such as X-rays, CT scans, and MRIs work much the same way: invaluable for spotting tumors, blockages, and structural damage, but usually only after disease has taken hold.
“Events that contribute to disease can happen at the molecular level, in milliseconds,” says Scott Fraser, Ph.D., president of imaging at Biohub. In type 2 diabetes, for example, pancreatic cells can compensate for dysfunction for years before blood sugar levels rise. To see the earliest signs of trouble, “we have to be able to scale from molecules to whole bodies — a billion-fold change in scale, and almost the same change in time.”
Further, even the most advanced microscopes leave much of biology unseen. “If we ask what fraction of the proteome [the full set of proteins active inside a cell] we can really look at, it’s probably less than a percent,” Fraser says. By combining multi-modal imaging with AI models that guide both data collection and image interpretation, Biohub is expanding what researchers can observe. New tools like laser phase plates, which enhance contrast in electron microscopy without increasing damage to cells, could push visibility from a narrow slice of cellular activity to more than half of the proteome.

Fraser says that in the next year or two, some of Biohub’s new imaging tools could go from something that’s exceptional to a tech that’s routine, and the coming decades could seem like sci-fi. When asked what medical imaging might look like 30 years from now, Fraser referenced the Star Trek tricorder — a hand-held health scanner — predicting they would become a reality, just as the smartphone once put supercomputers in our pockets. “You would discover not just that you’ve got a risk for something, but that you are starting to show signs. We’d be able to cure you before you even have symptoms.”
Tuning immune cells to individual needs
The human immune system isn’t optimized to protect us from the progressive diseases that hit us from midlife on. “The immune system evolved to only care about you until you’re about 20, because that would have been your reproductive age 200,000 years ago,” Califano explains. Tumors, for example, can exploit this limitation by co-opting immune cells that should attack them.
In a groundbreaking study in Cancer Cell, Califano and colleagues used gene editing to stop cancer-supporting T-cells from gathering in tumors in mice, spurring spontaneous remission. At Biohub, his team is building on this concept, using massive datasets and AI models to map the regulatory “logic” that governs how immune cells behave, identify genes that act as master switches, and eventually program a given individual’s immune system based on their unique needs. “You can embed logic into a computer so that it plays a video game, displays images, watches CNN, solves problems, and much more, right? You can do the same thing with a cell. There’s an almost infinite repertoire of things you could do,” Califano says.
Eventually, Califano envisions leveraging immune cells to detect early signs of disease, too. “We’ll use ‘reporters’ — innocuous cells that go in, explore your entire body, write a pathology report for you, then dissolve,” he says. “It’ll say, oh, you have this problem in the liver and this problem in the pancreas, but they can be fixed in these ways. The computer will come up with a recipe to implement that as effectively as possible.”
And in a couple of decades: a whole new class of therapies. “Now, you have a library of doctors. In the future, we will have libraries of cells — the cell that cures pancreatic cancer, the cell that cures renal cancer, the cell that cures Alzheimer’s. We could live not only much longer, but actually in much better condition, by reprogramming the immune system to [continually] rejuvenate the entire body.”
Mapping inflammation in real time
Researchers now know that at least half of deadly diseases — including heart disease and certain cancers — are driven in large part by inflammation, the body’s coordinated immune response to injury or threat. Yet the mechanisms that govern inflammation remain poorly understood, especially in its earliest stages.
“This is a hard problem, because inflammation results from the interaction of the immune system and healthy tissue within the body,” says Shana Kelley, Ph.D., president of bioengineering at Biohub. “We just don’t have technologies that would allow us to study inflammation in intact human tissue at the level of, let’s say, all of the proteins within our cells.”
Kelley’s team aims to fill that “white space” in research capabilities in a couple of ways. First, they’re putting engineered human tissues in petri dishes, promoting inflammation by introducing a wound, burn, or triggers for autoimmune disease, then collecting results on an ongoing basis. “Those large datasets are perfect material for the development of AI models that then can really point us towards factors of inflammation, or maybe even be key in the resolution of inflammation,” Kelley says.
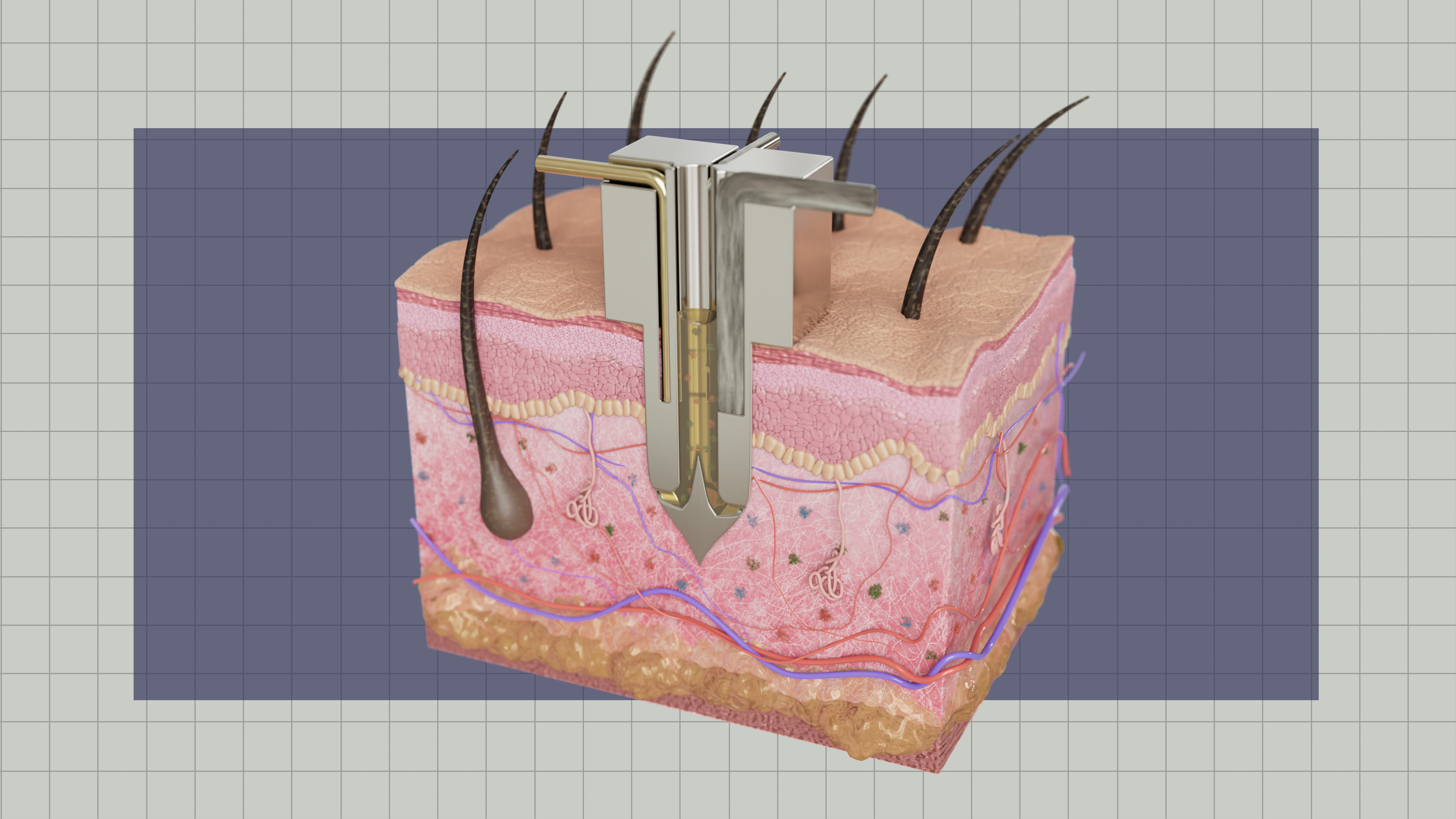
Second, the team is developing a device that can monitor inflammation from inside the body, similar to how a continuous glucose monitor tracks blood sugar levels. Last year, Kelley and colleagues published a paper on the first implantable inflammation sensor, which tracks cytokines, a standard marker. Another device in development will provide comprehensive analyses of skin inflammation, laying the groundwork for datasets, AI models, and detailed maps of inflammation.
The vision is to bring these pieces together — rich data sets made up of increasingly granular, molecule-level intel, along with the implantable monitors — into a system that could one day track inflammatory markers in real time and flag early signs of inflammation from within the body. “There would be the health benefit — fewer people suffering — and also economic benefits,” Kelley says. “It’s potentially transformational for society.”
This article was produced in partnership with Biohub, a nonprofit research organization developing AI-powered tools to accelerate and advance human health research. Visit biohub.org to learn more.